GIN Home
Ichneumonid Morphology
Subfamily Key
Lists of World Genera
Acaenitinae
Brachycyrtinae
Collyriinae
Lycorininae
Ophioninae
Poemeniinae
Rhyssinae
Stilbopinae
Xoridinae

By David B. Wahl & Ian D. Gauld
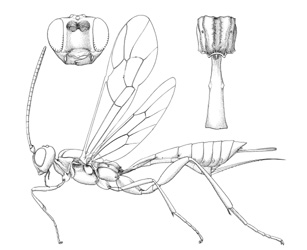
Collyria coxator |
Collyriines are rather rarely encountered wasps. Females are readily recognized by the unique ovipositor: long and slightly decurved, tapering to a sharp point, and with widely spaced denticles along the ventral margin.
Classification and diversity
The subfamily Collyriinae contains only eleven Palaearctic species in three genera, nine of which are in Collyria Schiødte (Yu & Horstmann, 1997; Gürbüz & Kolarov, 2006; Wahl et al., 2007; Sheng et al., 2012). The species are found from Spain through the Near East, and east to Mongolia and northern China; they often occur in rather open grassy areas, which have the highest diversity of their cephine hosts (Quinlan & Gauld, 1981). Collyria coxator has been introduced into the Nearctic for control of Cephus cinctus and C. pygmaeus; it has failed in the western Nearctic against C. cinctus but has been successfully established in the northeastern Nearctic as a parasitoid of C. pygmaeus (Shanower & Hoelmer, 2004).
The phylogenetic relationships of Collyria are presently unclear. Collyriines are morphologically highly autapomorphic and share few derived features with other subfamilies. This has resulted in them being placed by Gauld (1991) in his other subfamilies category, a group of subfamilies which are not clearly assignable to the Pimpliformes, Ophioniformes or other informal subfamilial grouping. A recent molecular study (Quicke et al., 2000) based on a single gene (the D2 region of 28s rDNA) has suggested that Collyria belongs in the Pimpliformes, but it lacks any of the adult or larval morphological synapomorphies of the clade (Wahl & Gauld, 1998). These observations suggest that the Collyriinae may be an isolated basal lineage of ichneumonids, highly specialized in attacking a small and otherwise seldom-utilized group of hosts.
The general biology of Collyriinae
All Collyria spp. appear to be egg-larval koinobiont endoparasitoids. Collyria coxator (Villers) (mostly recorded in the literature as calcitrator (Gravenhorst)), is a common parasitoid of the wheat-stem sawfly, Cephus pygmaeus (L.) in western Europe (Salt, 1931). Four other species of Collyria (catoptron Wahl, iberica Schmiedeknecht, orientator Aubert, trichophthalma (Thomson)) have been recorded as attacking hosts in the tribe Cephini (Cephus spp. and Trachelus spp.) (Shanower & Hoelmer, 2004). One exception is a host record of Janus integer (Norton) (Carlson, 1979), a species of Hartigiini that is a borer in the stems of Ribes (Grossulariaceae) spp. This single unconfirmed record requires confirmation.
A detailed account of the life-history of C. coxator is given by Salt (1931). Oviposition is into the relatively large egg of the cephine host, which is concealed within the tissue of its host food-plant stem.The female Collyria moves down the stem searching for the oviposition scars of the sawfly, and oviposits with her head facing downwards. The Collyria egg hatches before that of the host, with further development of the parasitoid taking place within the cephine larva. Larval development of the parasitoid is heteromorphic. The first and second instar larvae have large curved mandibles which presumably are used to eliminate competitors, including conspecific third instar larvae. A similar larval form was observed in C. catoptron, which is described (Wahl et al., 2007) as having a heavily sclerotized head capsule and sharp protuberances from the lateral part of the body. The first instar larva of C. coxator is free in the hosts haemocoel, but transformation to the second instar takes place in an evagination of the skin of the host. Subsequent larval instars of the collyriine are typically hymenopteriform. In late summer, the cephine larva is fully-grown and constructs its overwintering cocoon in the base of the grass stem. In early spring of the following year, the collyriine larva grows rapidly and consumes the entire cephine larva except for the cuticle. It then pupates within the hosts cocoon, not spinning one of its own.
Diagnosis
Small to medium-sized ichneumonids, fore wing length 5.0 to 6.8 mm. Labrum hidden. Clypeus weakly convex, to small, quadrate, and flattened; separated from supraclypeal area by epistomal suture; apex truncate, with small median tubercle. Malar space without subocular groove. Mandibular apex with two teeth. Antenna with flagellum filiform; males without tyloids. Gena long, about equal to length of eye in dorsal view. Ventral posterior corner of propleuron without strongly produced lobe. Epomia of pronotum absent. Notauli present, generally more or less meeting centrally. Sternaulus absent. Epicnemial carina present. Posterior transverse carina of mesothoracic venter absent. Propodeal carinae with transverse carina absent. Propodeal spiracle small and ovoid. Fore and middle tarsal claws with median tooth, hind tarsal claws simple. Fore tibia with dorsal apical tooth absent. Hind leg with coxa elongate, about 2x as long as width at midpoint; femur enlarged, about 3x as long as wide. Fore wing with areolet absent; vein 2m-cu with two bullae. Hind wing with vein 1/Rs > vein rs-m; veins 1/Rs, 1/M and 2/Cu tubular to spectral; vein 2/Cu about 0.4x as long as vein cu-a. Metasomal insertion on propodeum between hind coxae. Metasoma weakly laterally compressed. Metasomal segment 1 with T1 narrow anteriorly, posteriorly evenly broadened, with spiracles in front of midpoint; glymmae absent; anterior sclerotized part of S1 long, posterior end opposite spiracle; T1 and S1 free. Thyridium of T2 small and circular, immediately adjacent to anterior tergal margin. T2 with laterotergite separated by crease, laterotergite of T3 weakly separated, laterotergite of T4 pendant. T8+9 of female elongate. Hypopygium of female large and transverse, apical margin slightly produced medially. Ovipositor about as long as hind tibia; moderately decurved, compressed, rather deep basally and tapering to sharp cylindrical apex; margin of ventral valve with widely spaced denticles, dorsal valve without subapical notch.
The Collyriinae present in the Nearctic
Collyria Schiødte, 1839 (HOD)
· Collyria coxator (Villiers), 1789 (HOD)
Identification and resources
Females of Collyria coxator are easily recognized. The ovipositor is weakly curved downward, tapering from base to apex, with small weak teeth on ventral margin. In addition, T8+9 is elongate. Males are more difficult to identify, but the following characters should suffice: a) the apical margin of the clypeus has a small median tubercle; b) the gena is about as long as the eye; c); the propodeum lacks transverse carinae; d) the fore wing lacks an areolet; e) the hind wing has vein 1/cu about 0.3x as long as vein cu-a; f) metasomal segment 1 is elongate and narrow in dorsal view, with the glymma absent and the spiracle a little in front of the middle.
Hymenoptera Online Database (HOD)References
Carlson, R.W. 1979. Family Ichneumonidae. In: Krombein, K.V., P.D. Hurd, D.R. Smith, and B.D. Burks (eds.). Catalog of Hymenoptera of America North of Mexico. Vol. 1. Smithsonian Institution Press: Washington, D.C. 1198 pp.
Gürbüz, M.F. & Kolarov, J. 2006. A review of the Collyriinae (Hymenoptera: Ichneumonidae). Entomologica Fennica 17: 118-122.
Kuslitzky, V.S. & Kasparyan, D.R. 2011. A new genus of ichneumonid flies of the subfamily Collyriinae (Hymenoptera: Ichneumonidae) from Syria and Israel. Zoosystematica Rossica 20: 319-324.
Gauld, I.D. 1991. The Ichneumonidae of Costa Rica, 1. Memoirs of the American Entomological Institute, 47: 1-589.
Quicke, D.L.J., Fitton, M.G., Notton, D.G., Broad, G.R. & Dolphin, K. 2000. Phylogeny of the subfamilies of Ichneumonidae (Hymenoptera): a simultaneous molecular and morphological analysis, pp. 74-83. In: Austin, A.D. & Dowton, M, (eds.) Hymenoptera. Evolution, Biodiversity and Biological Control. CSIRO Publishing: Collingwood, 468 pp.
Quinlan, J. & Gauld, I.D. 1981. Symphyta (except Tenthredinidae). Handbooks for the Identification of British Insects 6(2)(a): 1-67.
Salt, G. 1931. Parasites of the wheat stem sawfly, Cephus pygmaeus Linnaeus, in England. Bulletin of Entomological Research, 22: 479-545.
Shanower, T.G. & Hoelmer, K.A. 2004. Biological control of wheat stem sawflies: past and future. Journal of Agricultural and Urban Entomology, 21: 197-221.
Sheng, M.L., Broad, G.R., & Sun, S.P. 2012. A new genus and species of Collyriinae (Hymenoptera, Ichneumonidae). Journal of Hymenoptera Research 25: 103-125.
Wahl, D.B. & Gauld, I.D. 1998. The cladistics and higher classification of the Pimpliformes (Hymenoptera: Ichneumonidae). Systematic Entomology, 23: 265-298.
Wahl, D.B., Shanower, T.G. & Hoelmer, K.A. 2007. A new species of Collyria Schiødte (Hymenoptera: Ichneumonidae: Collyriinae) in China, and potential biological control agent for Cephus cinctus in North America. Journal of the Kansas Entomological Society, 60: 43-50.
Yu, D.S. & Horstmann, K. 1997. A catalogue of World Ichneumonidae. Part 1: Subfamilies Acaenitinae to Ophioninae. Memoirs of the American Entomological Institute 58 (1): 1-763.
Site credits and citation. Questions? E-mail: aei[at]aei.cfcoxmail.com